表面增强红外光谱(SEIRA)学中的多分子层协同化学吸附现象
何林涛 Peter R. Griffiths
Department of Chemistry, University of Idaho, Moscow, ID83844-2343, USA
(*已发表在光谱学与光谱分析,18,3,1998)
Osawa[1]曾建议表面增强红外光谱(SEIRA)效应来自于电磁机理和化学吸附机理的贡献,类似于表面增强拉曼光谱(SERS)效应。通过比较邻、间、对-硝基苯甲酸在Ag表面上的表面增强红外光谱,Osawa等人[2]阐明了SEIRA的表面增强选律:只有偶极矩变化方向或其分量垂直于金属表面的振动模 式才是SEIRA活性的。
本文报导了以显微透射 SEIRA测量方式,在高真空条件下,2,4-二硝基苯甲酸在银表面的多分子层协同化学吸附现象。实验结果表明2,4-二硝基苯甲酸和3,5-二硝基苯甲酸的COOH和NO2反对称振动模式在其SEIRA光谱具有活性,我们推测底层分子是以垂直取向吸附在银表面,而上层分子则以倾斜方向排列。图1是在高真空情况下不同的时间,2,4-二硝基苯甲酸在银膜表面的SEIRA光谱。
利用SEIRA模型化合物对-硝基苯甲酸(PNBA),我们也观察到多分子层协同化学吸附和溶剂效应对PNBA的SEIRA光谱的影响,在PNBA的多分子层SEIRA光谱中,COO-对称伸缩振动峰分裂成双重峰,因为上层的-COO-官能团与银表面的相互作用较底层分子弱,我们将1420cm-1峰归属于上层PNBA的COO-对称伸缩振动,1387cm-1为相应的下层PNBA 的COO- 振动。比较溶剂丙酮和甲醇对PNBA在银表面形成薄膜时的影响,当使用甲醇作溶剂时,1420cm-1峰要比1387cm-1强得多。当PNBA溶液在银表面的吸附与脱附伴随着溶剂挥发的过程中,因为甲醇与PNBA分子的强氢键作用,导致更多的PNBA-离子处于上层分布,以倾斜取向吸附在银表面,因此,从甲醇溶液形成的PNBA SEIRA光谱,其上层COO-伸缩振动峰(1420cm-1)相对于使用丙酮溶液时强得多。图2显示了对-硝基苯甲酸在不同溶剂中的SEIRA光谱。
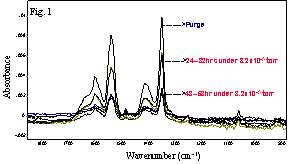
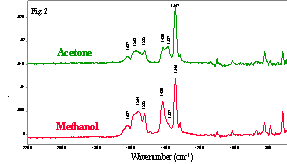
Fig.1. SEIRA spectra of 2,4-dinitrobenzoic acid on 5 nm Ag / ZnSe at 8.2x10-5 torr during the period from 0 to 53 hours after deposition.
Fig.2. SEIRA spectra of multilayers of
p-nitrobenzoic acid on 5 nm Ag / ZnSe after deposition from acetone solution
(above) and methanol solution (below).
Multilayer Cooperative Chemisorption in Surface-
Enhanced
Infrared Absorption (SEIRA) Spectroscopy
Lin-Tao HE and
Peter R. Griffiths
Department of
Chemistry, University of Idaho, Moscow, ID 83844-2343
(* Published in
Proc. 11th Int. Conf. Fourier Transform Spectrosc.,Am. Inst. Phys.
Conf. Proceedings 602, 1997)
Osawa has suggested
that both electromagnetic and chemical mechanisms contribute to the total
magnitude of the enhancement of vibrational bands in surface-enhanced infrared
absorption (SEIRA), in a similar manner to surface-enhanced Raman scattering
(SERS). Comparing the SEIRA
spectra of o, m, and p-nitrobenzoic acid adsorbed on Ag films with only those
vibrational modes whose dipole
derivative has a component that is perpendicular to the metal surface being
SEIRA active, Osawa’s group demonstrated the surface selection rule known for reflection-absorption
spectroscopy of adsorbates on the smooth metal surface also holds for SEIRA
spectroscopy.
In this paper, we
will report the cooperative chemisorption of multilayers of 2,4-dinitrobenzoic
acid on a silver surface under high vacuum studied by microtransmission
spectrometry. The fact that the antisymmetric stretching modes of carboxylate
and nitro groups are active in SEIRA spectra of 2,4- and 3,5-dinitrobenzoic
acid suggests that the bottom layer is adsorbed perpendicular to the metal
surface with higher layers oriented obliquely.
Using
p-nitrobenzoic acid (PNBA) as a model compound, we also observed the multilayer
cooperative chemisorption and the effect of the solvent on the SEIRA spectra of
PNBA. The symmetric COO- stretch in the SEIRA spectra of multilayers
of PNBA is split into a doublet. Because there is a weaker interaction between
COO- group of PNBA in the upper layers with the Ag surface than for
the bottom layer, we assign the 1420 cm-1 component to the symmetric
COO- stretch of the upper layers of PNBA and the 1387 cm-1
band to the corresponding mode of the lower layer. When the films are cast from
methanol and acetone, it is found that the band at 1420 cm-1 is much
stronger than the peak at 1387 cm-1 when the film is cast from
methanol. Because of the effect of hydrogen bonding between methanol and PNBA
molecules during the period of deposition and adsorption of PNBA solution on
the Ag surface with solvent evaporation, more PNBA- species from the
methanol solution exist at oblique orientations on the Ag surface than when
acetone is used as the solvent.
参考文献
1. Osawa, M., and Ikeda, M., J. Phys. Chem. 95, 9914-9919 (1991).
2. Osawa, M., Ataka, K-I., Ikeda, M., Uchihara, H.,
Nanba, R., Anal. Sci. 7,
503-506 (1991).